
Restylane® Defyne is also indicated for injection into the mid-to-deep dermis (subcutaneous and/or supraperiosteal) for augmentation of the chin region to improve the chin profile in patients with mild to moderate chin retrusion. Restylane® Defyne is for mid-to-deep injection into the facial tissue for the correction of moderate to severe deep facial wrinkles and folds, such as nasolabial folds. Restylane® Refyne is for mid-to-deep injection into the facial tissue for the correction of moderate to severe facial wrinkles and folds, such as nasolabial folds. Restylane® Kysse is for lip augmentation and for correction of upper perioral wrinkles. Restylane® Silk is for lip augmentation and for correction of perioral wrinkles. Restylane® Lyft with Lidocaine is also indicated for injection into the dorsal hand to correct volume loss. Restylane® Lyft with Lidocaine is for deep implantation into the facial tissue for the correction of moderate to severe facial wrinkles and folds, such as nasolabial folds and for cheek augmentation and for the correction of age-related midface contour deficiencies. Restylane® and Restylane-L® are also indicated for injection into the lips. Restylane® and Restylane-L® are for mid-to-deep injection into the facial tissue for the correction of moderate to severe facial wrinkles and folds, such as nasolabial folds. The Restylane family of products are indicated for patients over the age of 21, and includes Restylane®, Restylane-L®, Restylane® Lyft with Lidocaine, Restylane® Silk, Restylane® Kysse, Restylane® Refyne, Restylane® Defyne, and Restylane® Contour. Restylane Contour Product & Safety Information Restylane Defyne Product & Safety Information Restylane Refyne Product & Safety Information 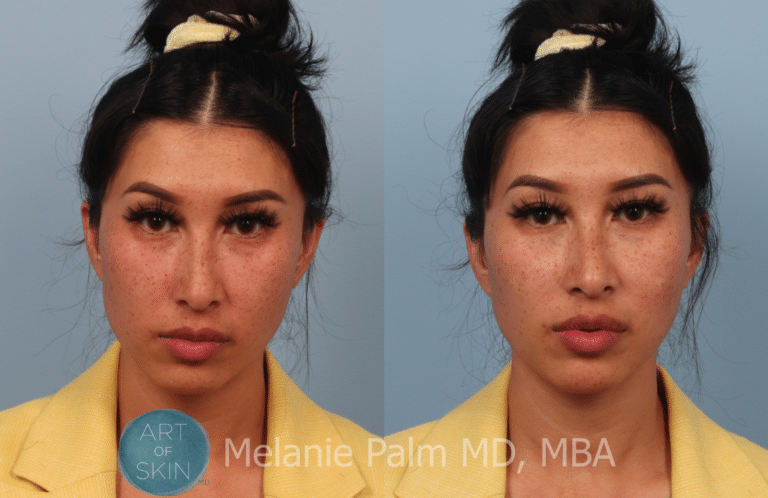
Restylane Lyft Product & Safety Information Restylane Silk Product & Safety Information Restylane Kysse Product & Safety Information Fort Worth, TX: Galderma Laboratories, L.P., 2016. Systematic review of clinical trials of small- and large-gel-particle hyaluronic acid injectable fillers for aesthetic soft Accessed on May 18, 2018.Ĭohen JL, Dayan SH, Brandt FS, et al. MedicalDevices/ ProductsandMedicalProcedures/ DeviceApprovalsandClearances/ Recently-ApprovedDevices/ ucm609592.htm. Medical Devices, Restylane Lyft with Lidocaine - P040024/S099. Restylane CONTOUR Instructions for Use. 
Evaluation of rheometry amplitude sweep cross-over point as an index of flexibility for HA fillers. Perceived naturalness of facial expression after hyaluronic acid filler injection in nasolabial folds and lower face. Philipp-Dormston WG, Schuster B, Podda M.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
